 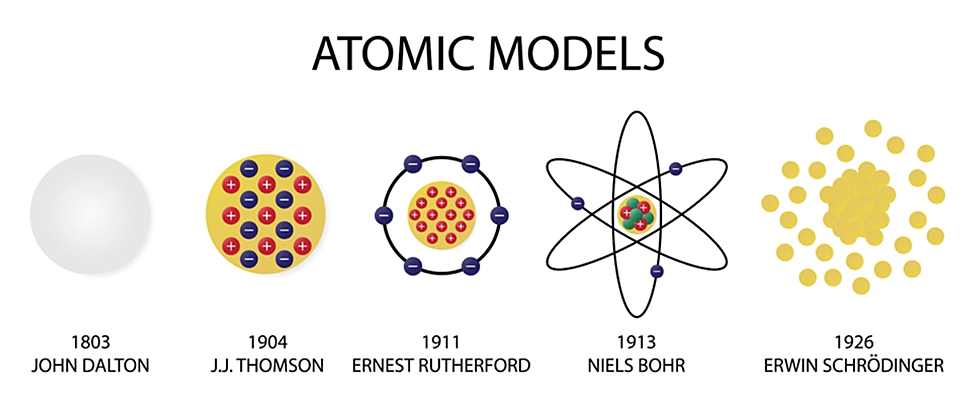 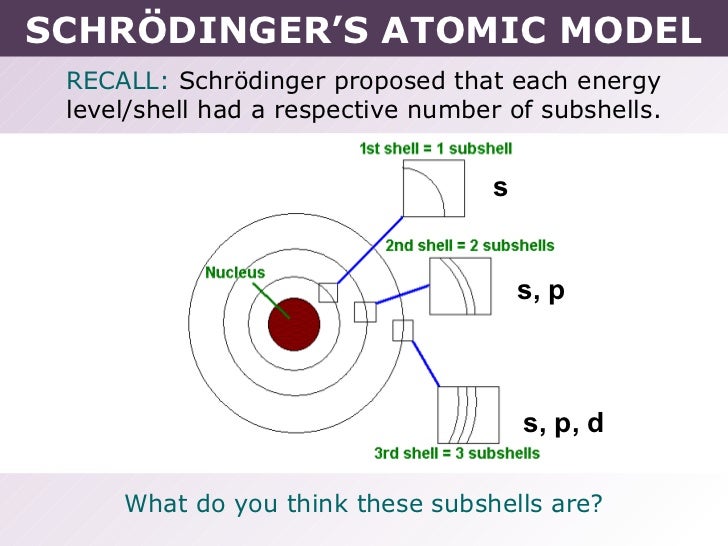
He won the 1933 Nobel prize in physics with colleague Paul Adrien Maurice Dirac "for the discovery of new productive forms of atomic theory". The Viennese scientist Erwin Schrödinger became world famous at a relatively late age, that is, for a theoretical physicist active during the 1920’s. Schrödinger proposed an original interpretation of the physical meaning of the wave function. He won the 1933 Nobel prize in physics with colleague Paul Adrien Maurice Dirac "for the discovery of new productive forms of atomic theory" Erwin Rudolf Josef Alexander Schrödinger, sometimes written as Erwin Schrodinger or Erwin Schroedinger, was a Nobel Prize-winning Austrian physicist who developed a number of fundamental results in the field of quantum theory, which formed the basis of wave mechanics: he formulated the wave equation (stationary and time-dependent Schrödinger equation) and revealed the identity of his development of the formalism and matrix mechanics. For this discovery, Erwin Schrödinger was awarded the 1933 Nobel Prize in Physics.Erwin Rudolf Josef Alexander Schrödinger, sometimes written as Erwin Schrodinger or Erwin Schroedinger, was a Nobel Prize-winning Austrian physicist who developed a number of fundamental results in the field of quantum theory, which formed the basis of wave mechanics: he formulated the wave equation (stationary and time-dependent Schrödinger equation) and revealed the identity of his development of the formalism and matrix mechanics. In a surprising fashion, however: Schrödinger demonstrated that electrons could have the properties of either waves or particles, but are neither the one nor the other their state can be calculated only with a degree of probability. The dispute as to whether quantum objects such as electrons, atoms or molecules were waves or particles was settled. In this paper, he first formulated his famous wave equation, which has gone down in the annals of physics as the “Schrödinger equation.” The wave equation makes it possible to calculate the energy levels of electrons in an atom, thus solving one of the great problems in quantum physics.Īfter Schrödinger’s wave equation, nothing in the world of physics was the same again. Erste Mitteilung” (Quantization as a problem of proper values, part one), which he sent to the Annalen der Physik on 26 January 1926. These efforts resulted in his first article, “Quantisierung als Eigenwertproblem. This vacation was the beginning of his annus mirabilis, a phase, lasting some twelve months, of concentrated, creative work. What were the properties of such waves of matter? Schrödinger spent Christmas and New Year 1925/26 studying the matter while on holiday in Arosa. Schrödinger focused intensively on Broglie’s proposition that all matter has wave properties. This contradicted the prevailing opinion of leading physicists of the time, who assumed that electrons were particles. In the summer semester of 1925, Erwin Schrödinger had read the doctoral thesis of a young Frenchman, Louis de Broglie, who proposed that matter – such as electrons – also possessed wave properties. The simplest form of the Schrödinger Equation is as follows: H E (2.2.1) (2.2.1) H E. It is a mathematical equation that defines the electron’s position, mass, total energy, and potential energy.  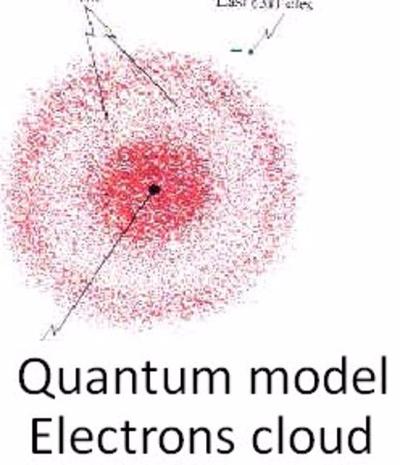
While at the University, Erwin Schrödinger revolutionized physics by creating a new atomic theory, a scientific breakthrough he achieved in the winter of 1925/26. Schrödingers Equation describes the behavior of the electron (in a hydrogen atom) in three dimensions. At that time no one imagined that six years later he would leave the University and the city hailed as a genius by luminary figures such as Albert Einstein and Max Planck, and celebrated as a star. In the fall of 1921, Erwin Schrödinger was appointed to the chair for theoretical physics at the University of Zurich, a position that had been vacant since 1914. Erwin Schrödinger proved that electrons could have the properties of either waves or particles, but are neither the one nor the other – a discovery that revolutionized physics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
